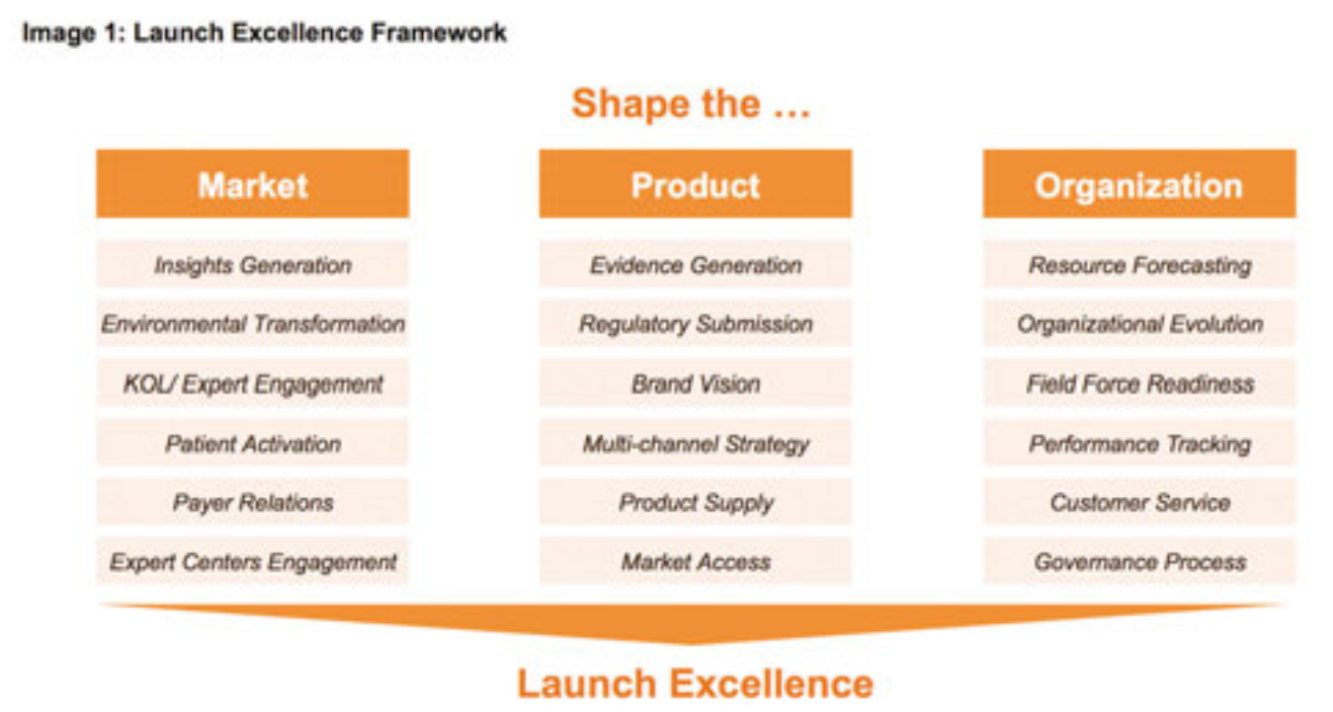
The importance of a pharma product’s launch to its success cannot be overstated. A recent analysis by Deloitte showed that most new drugs continue with the revenue trajectory set at launch – around 70% of products that miss expectations at launch continue doing so in subsequent years, and around 80% of products that meet or beat expectations continue to do so afterward.
In other words, you’ve got one shot to get it absolutely right. And with ever-increasing development costs and in an ever-more competitive landscape, the need to get it right is greater than ever. But it’s more complex these days, too. Whether it’s more specialty products, evolving regulatory frameworks, increasingly diverse territories, increasing payer influence or the need for multichannel communication, the complexity of a global product launch is only increasing. COVID-19 – and the subsequent demise of traditional face-to-face sales representative time – has only added to the list of challenges.
Many companies are struggling to get it right. According to a recent report by L.E.K. Consulting, half of drugs rolled out since 2004 didn’t live up to their sales forecasts. And according to IQVIA, fewer than 10% of launches are categorised as ‘excellent’.
So why does it go wrong so often? There are many potential reasons, including a lack of engagement with key medical and payer stakeholders, an inadequate pricing and market access strategy or an overstated valuation by the company. But while some errors are unforeseen, many can be prevented with better planning, coordination, communication and transparency in launch planning and implementation.